OUT OF EQUILIBRIUM PHYSICAL CHEMISTRY AND NANOMATERIALS
The non-covalent assembly of nano-objects in solution, from polymers, surfactants, nanoparticles or proteins has been studied in our group, with emphasis on the relationship between the aggregate morphology and the chemistry of the elementary bricks. The goal is to generate hierarchical structures in the nano- and micro- domains for applications in the field of nanoscience. Recently we investigated the impact of thermodynamic processes and transformations that accompany assembly mechanisms using Isothermal Titration Calorimetry (ITC).
The thermodynamics of electrostatic assemblages was studied in various contexts, e.g. on synthetic macromolecules, on a system relevant in the natural aquatic environment and on the surfactant deposition on cotton fibers. These studies have revealed that electrostatic interaction between oppositely charged species is entropically drive, and associated with the release of the bound counterions.
REFERENCES
E.K. Oikonomou*, N. Christov, G. Cristobal, C. Bourgaux, L. Heux, I. Boucenna and J.-F. Berret*
Design of eco-friendly fabric softeners: structure, rheology and deposition on micro- and nanocellulose fibers
Journal of Colloid and Interface Science 525, 206–215 (2018)
https://doi.org/10.1016/j.jcis.2018.04.081
F. Loosli , L. Vitorazi, J.-F. Berret and S. Stoll*
Towards a better understanding on agglomeration mechanisms and thermodynamic properties of TiO2 nanoparticles interacting with natural organic matter
Water Research 80, 139 – 148 (2015)
https://doi.org/10.1016/j.watres.2015.05.009
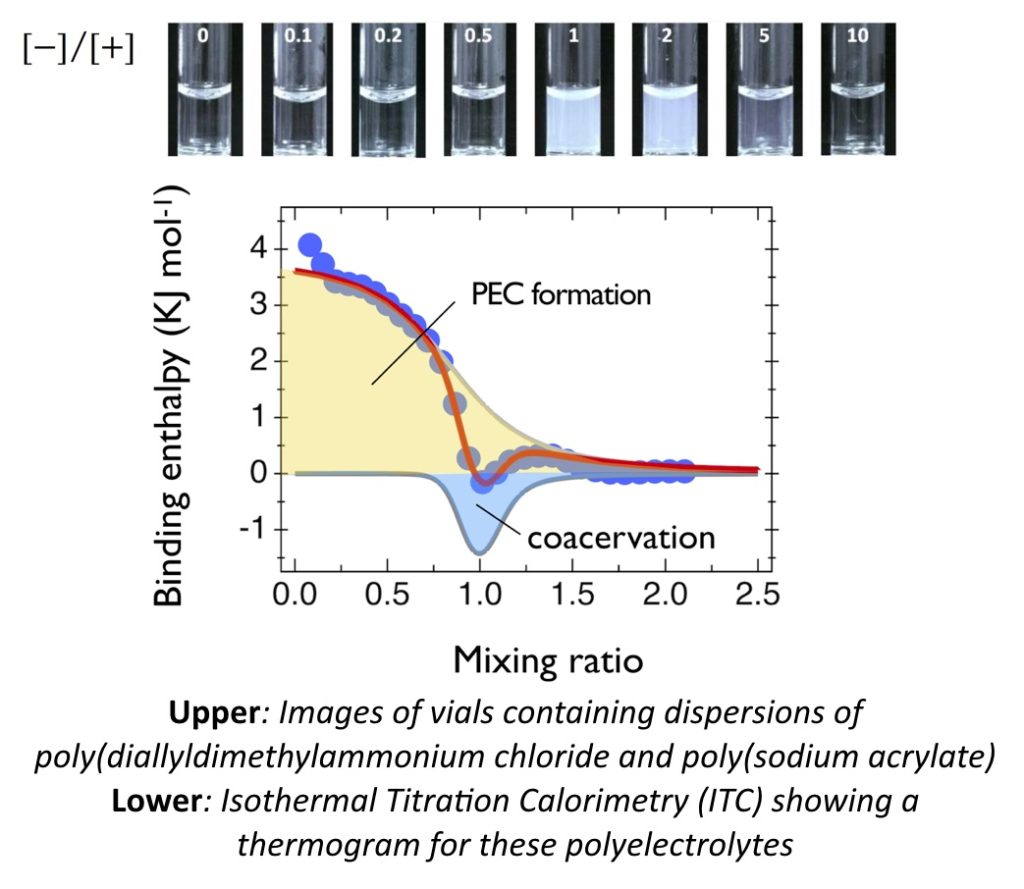
L. Vitorazi, N. Ould-Moussa, S. Sekar, J. Fresnais, W. Loh, J.-P. Chapel and J.-F. Berret*
Evidence of a two-step process and pathway dependency in the thermodynamics of poly(diallyldimethylammonium chloride)/poly(sodium acrylate) complexation
Soft Matter 10, 9496 – 9505 (2014)
https://doi.org/ 10.1039/C4SM01461H